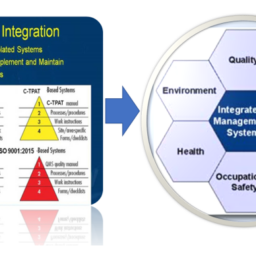
What is Laboratory Management based on ISO 17025:2017 Accreditation exactly?
Laboratory management according to the ISO 17025 standard aims to secure the results of tests or calibrations as well as the technical credibility and competence of laboratories.
If you are a laboratory, you can implement a quality management system (also called QM system) for this purpose. The relevant standard for quality management in laboratories is DIN EN ISO/IEC 17025:2017. It places requirements on the competence of laboratories.
Unlike other management standards, laboratory management is not primarily concerned with meeting customer requirements. Instead, the focus is on the quality and reliability of your testing and calibration methods. In addition to the general requirements for laboratory management, the standard also defines the technical requirements for a laboratory. Accordingly, the laboratory must consider and comply with the important factors that determine the reliability and accuracy of the tests. After the successful implementation of a QM system, it can then be accredited.
Accreditations are now based on ISO 17025:2017. The previous version of the standard was ISO 17025:2005. During accreditation, the implementation of the implemented quality management system and the specific requirements for technical competence are checked by a Notified Accreditation Body. The audit primarily includes the staff who manage the laboratory, the methods and procedures used, the validation of the methods, the participation in and implementation of proficiency tests (interlaboratory comparisons), the traceability of the results generated and the correct use of suitable facilities and equipment used to generate the results be used.
Why is ISO 17025 accreditation important for your laboratory management?
An accredited quality management system according to ISO 17025 is not mandatory for laboratory management. Nevertheless, accreditation offers you as laboratory various advantages. Today, this is often a prerequisite for the recognition of the services provided by testing or calibration laboratories. The laboratories are conformity assessment bodies (KBS), which, with the help of specialist knowledge, ensure the verifiable correctness of the results generated in the laboratory and documented on the test reports or calibration certificates. The impartial generation and traceability of the results generated in the laboratory is particularly important. This is indispensable from a qualitative or legal point of view or in the event of possible liability claims. Accredited QM systems show the client that it is a competent, independent and trustworthy laboratory. A national, named accreditation body certifies this and regularly monitors the accreditation through assessments. In the U.S., for example, these are organizations like Perry Johnson Laboratory Accreditation (PJLA), or DNV, BSI, etc…
In addition, laboratory accreditation offers the following advantages, among others:
- Improving performance through clearly regulated processes
- Increase in quality awareness among employees
- Advantage over non-accredited competitors
How do you prepare for accreditation in laboratory management?
The accreditation process for your laboratory according to ISO 17025 should be as efficient as possible. You should make some preparations for this. First, you will take stock of your operational processes and facility. In addition, all existing procedures and documents are viewed. This is followed by the actual project planning. The members of the project team are determined and deadlines and measures are planned.
The next step is to create conditions for successful accreditation. This involves, for example, laboratory organization, flawlessly functioning technology and the distribution of information and appropriate training. This also includes the creation of important documents. Examples are the QM Manual, corresponding work instructions and control plans. The necessary test and calibration instructions and the associated device documentation must also be created. The methods and procedures are then verified and validated.
Now your quality management system can be introduced. It is important that you train your employees accordingly and make them aware of the importance of the system. Because only if you live the QM system in practice it can bring lasting success. It is also important to continuously improve and monitor the effectiveness of your system. This can be achieved using internal audits.
Finally, you can apply for the accreditation process. After you have sent all the necessary documents to the Accreditation Body, the accreditation audit will be carried out on your premises.
What is the difference between an accreditation and a certification?
The definition for the term accreditation is the “confirmation by a third party that formally demonstrates that a conformity assessment body has the ability to carry out specific conformity assessment tasks”. Thus, an accreditation is the confirmation of the competence to carry out certain tasks. In contrast to certification, for example according to ISO 9001, according to ISO/IEC 17025 only the test and calibration methods listed on the issued certificate and in its appendix are accredited. The accreditation ensures that the results are accepted internationally and by authorities without re-examination.
How does the accreditation process work?
The course of an accreditation procedure by the Accreditation Body is divided into four steps. It starts with the application process, then an assessment takes place, followed by the actual accreditation and finally there is monitoring. Below you will find a brief explanation of all the steps.
Step 1 – Application for Accreditation
The accreditation process begins with your application for accreditation. It is necessary to submit the application formally. Make sure that all required documents are enclosed with the application.
We at JJK Consulting & Auditing can help you out with orientation on that process. Or you can find these on the Accreditor’s website. Finally, the Accreditation Body checks your application for completeness. You will also be informed of a contact person who is responsible for the further accreditation process. If desired, you can also arrange a preliminary inspection prior to the actual assessment. However, this is not free of charge.
Step 2 – Assessment
The assessment takes place in the second step of your accreditation procedure. First of all, the responsible assessors are selected. In addition, the submitted documents are checked and the scope of the assessment is determined. If there are no deviations, the assessment can take place on site. An assessment plan will be sent to you in advance. After the on-site assessment, follow-up work takes place. Reports are created for assessment and analyzes are carried out for any deviations. On-site follow-up assessments are also possible.
Step 3 – Accreditation
The third step includes the actual accreditation, i.e. the decision as to whether all requirements and competencies have been met. This decision is made by the accreditation committee. You will then receive a written notification. If the decision is positive, you will also receive an accreditation certificate. In the event of a negative decision, the notification contains a corresponding justification. After successful accreditation, you will be included in the directory of accredited bodies and can make them public.
Accreditation process
Step 4 – Monitoring and Change
It is then regularly checked whether the requirements are still being met. So-called monitoring assessments are carried out. If deviations are found, appropriate corrective measures must be taken. If these cannot be remedied by measures, the accreditation can be restricted, suspended or revoked. The intervals for such surveillance assessments are specified for each accreditation activity. After five years, a re-accreditation takes place.

JJK Consulting & Auditing offers ISO 17025:2017 Gap assessment audits and training on quality management in the laboratory. Our experts can certainly make you fit for the introduction or ongoing further development of your laboratory management system regarding ISO17025.
With our step-by-step implementation methods, you can quickly fill your knowledge gaps on the entire range of DIN ISO IEC 17025.
Our experts are all ISO17025:2017 certified Lead Auditors with many years of implementation experience.
BECOME ISO17025:2017 ACCREDITED ON TIME & BUDGET
JJK Consulting offers everything you need to implement an ISO 17025:2017 LMS, you don’t need to go anywhere else. We guarantee Accreditation for your company or organization (provided you follow our advice!). You benefit from real-world practitioner expertise, not just academic knowledge.
We offer ISO 17025:2017 – Implementation – Audit Service – Training – Services available in the NY Metro Area and Nationwide Onsite or within Remote/VPN secure teleconference meetings.
Ready to Explore New Possibilities?
 Please contact JJK Consulting at your convenience for an initial consultation on ISO 17025:2017 Accreditation.
Please contact JJK Consulting at your convenience for an initial consultation on ISO 17025:2017 Accreditation.
We’ll work with you one on one to determine a roadmap to success.
Call Today: 973-402-5889
#iso17025 accreditation